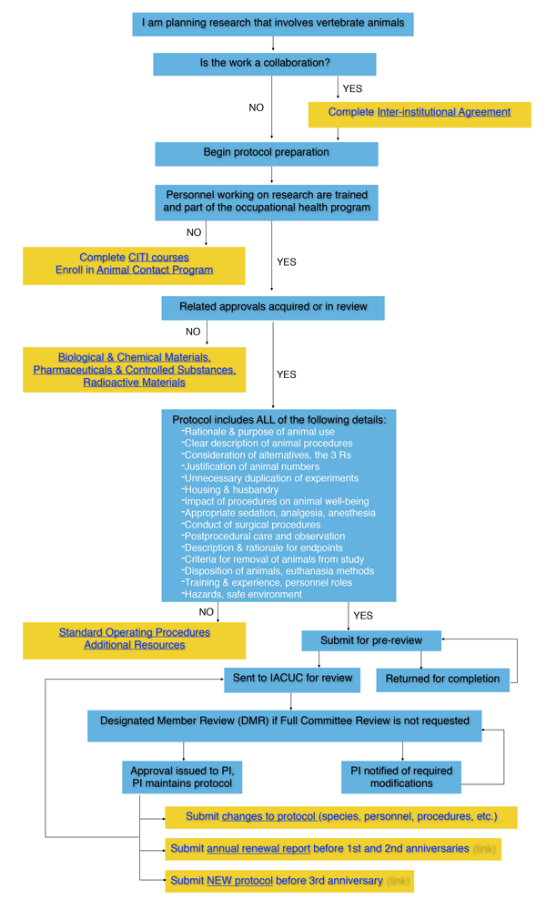
I am planning research that involves vertebrate animals.
- Is this a work collaboration?
- YES
- NO
- Go to Step 2
- Begin Protocol Preparation
- Personnel working on research are trained and part of the occupational health program?
- YES
- Go to Step 4
- NO
- Complete CITI courses
- Enroll in Animal Contact Program
- YES
- Related approvals acquired or in review?
- Protocol includes ALL of the following details:
- Rationale & purpose of animal use
- Clear description of animal procedures
- Consideration of alternatives, the 3 Rs
- Justification of animal numbers
- Unnecessary duplication of experiments
- Housing & husbandry
- Impact of procedures on animal well-being
- Appropriate sedation, analgesia, anesthesia
- Conduct of surgical procedures
- Postprocedural care and observation
- Description & rationale for endpoints
- Criteria for removal of animals from study
- Disposition of animals, euthanasia methods
- Training & experience, personnel roles
- Hazards, safe environment
- YES
- Go to Step 6
- NO
- Submit for pre-review
- Returned for completion
- Complete and re-submit for pre-review
- Sent to IACUC for review
- Proceed to Step 7
- Returned for completion
- Designated Member Review (DMR) if Full Committee Review is not requested
- PI notified of required modifications
- Repeat step 7
- Repeat step 7
- Approval issues to PI, PI maintains protocol.
- Submit Changes to Protocol (species, personnel, procedures, etc.) to IACUC for review
- Submit Annual Renewal Report before 1st and 2nd anniversaries to IACUC for review
- Submit New Protocol before 3rd anniversary to IACUC for review
- PI notified of required modifications

 Give to Florida Tech
Give to Florida Tech 